Application process
The NBB has an open access policy and annually receives and reviews 100-150 requests from academic researchers and pharmaceutical companies from all over the world. The NBB follows standard procedures to ensure an effective organization of the tissue transfer process.
Preparing the request for samples
The NBB’s extensive stock of human CNS tissue samples are registered in a members-only online database: the e-NBB. New members can request an account and after approval they can search the collection and make and save a selection of brain samples.
If you need help in selecting samples, please contact the NBB via eNBB@nin.knaw.nl. Discussion between applicants and the NBB is encouraged prior to submission of the application. The personnel of the NBB may be able to provide useful advice and expertise on the use of human tissue and tissue availability. Applicants may need to modify their application before submission, dependant on diagnosis, region and amount of tissue available.
Submission and review of the request
You can request samples by completing one of the application forms below and sending it to eNBB@nin.knaw.nl, accompanied by the e-NBB sample selection (optional).
Application form for non-profit organizations
Application form for pharmaceutical organizations (for-profit)
Supplementary application form (for requesting additional tissue for a project that has been approved earlier)
All applications are reviewed by the NBB’s scientific committee. The review process is based on the availability of the material (diagnosis and anatomical area) and scientific quality of the project. Within 6 weeks upon receipt of the application form, we aim to inform you whether your request can be approved. The document linked below describes the review criteria for applications.
Review criteria for tissue applications
Approval and financial contribution
Once an application has been approved, a project number is assigned and the researcher is informed about the subsequent procedure for obtaining the requested samples and the financial contribution. In general, for non-profit organizations, this is € 70 per tissue block, 2 ml CSF or plasma, or ten FFPE tissue sections, € 35 for a DNA sample, and € 150 for a sample of microglial cells.
The NBB operates a different pricing model for for-profit organizations; estimates can be provided upon request. When submitting grants for research projects which plan to use NBB tissue, we recommend to include the estimated financial contribution in the grant application budget.
In recent years we have seen a significant increase in project applications from public-private consortia. Considering the difficult task of determining a non-profit or for-profit character of these applications, and the lack of information on ownership and sharing of data and intellectual property within these collaborations, the NBB has decided to add a third cost-reimbursement category: public-private partnerships. These are defined as projects on initiative of the academic partner where all or part of the project costs are carried by industry, whereas the project execution lies predominantly within academia. This will maintain our desire to remain accessible to academic researchers, while recognizing the for-profit character and interests of the funding source.
Invoice Information
An invoice for the supplied samples will be sent after the samples have been received by the applicant. If the NBB has not supplied samples to the applicant's organisation before, 25% of the total financial contribution shall be invoiced in advance.
Please read more about our invoicing information and payment conditions here.
The NBB is a non-profit organization and the financial contribution is meant as compensation for the costs of brain banking, and not as payment for the brain tissue samples as such. Costs like donor recruitment, autopsies around the clock, processing samples in the lab, determining the neuropathological diagnosis, and handling and storing samples are very high and the NBB is almost completely dependent upon grants and donations.
Material Transfer Agreement
The NBB requires a signed Material Transfer Agreement (MTA) to ensure that all conditions for the transfer and the use of the material are agreed upon by the receiving and providing parties. The MTA must be signed by an authorized representative of the university or institute in question. One master MTA per research institute (legal entity) covers all legal terms of delivery. Based on this MTA, all researchers in the research institute may apply for tissue.
 After approval of an application, a Material Transfer Statement (MTS; a clear summary of the MTA), is sent to the researcher along with an Implementing Letter. This letter of approval specifies the material that will be supplied and the financial contribution. Upon receipt of the Implementing Letter and MTS, both signed by the applicant, the material will be supplied. The main applicant then also receives the extensive (anonymized) clinical and neuropathological information of the donors from which samples have been supplied. The clinical summary contains, among other data, the cause of death and the medical history (e.g. clinical course and medication use).
After approval of an application, a Material Transfer Statement (MTS; a clear summary of the MTA), is sent to the researcher along with an Implementing Letter. This letter of approval specifies the material that will be supplied and the financial contribution. Upon receipt of the Implementing Letter and MTS, both signed by the applicant, the material will be supplied. The main applicant then also receives the extensive (anonymized) clinical and neuropathological information of the donors from which samples have been supplied. The clinical summary contains, among other data, the cause of death and the medical history (e.g. clinical course and medication use).
Transportation
The transportation method depends on the storage type(s) of the selected tissue. Please note that frozen tissue samples are shipped on dry ice, which means that they are exposed to CO2, despite the fact that the samples are sealed in aluminum vials or plastic bags. This causes a temporary drop in pH, which results in changes to the properties of the proteins in the sample. Therefore, depending on the type of analysis to be performed on the sample, recipients may need to allow the samples to “normalize” after being removed from the shipment container. Murphy et al. (2013) describe two ways to do this: the first is allowing the samples to normalize in a freezer for 96 hours prior to performing the assay, to allow the CO2 to dissipate. If it is not possible to wait for 96 hours, a second solution is to remove the cap and vent the CO2 before thawing the sample. A summary of the article by Murphy et al. can be found here.
Summary of the material transfer process
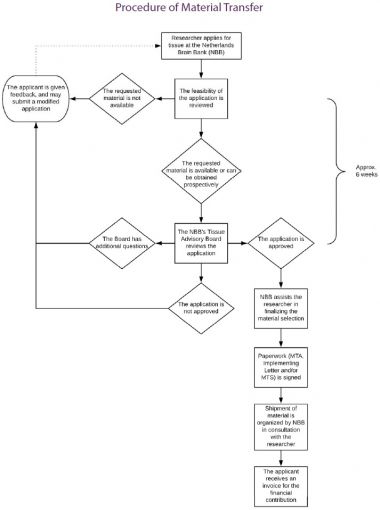
- The researcher employed by or affiliated with a certain Legal Entity, such as a university or a pharmaceutical company, sends an Application Form to the NBB.
- The Application Form is reviewed by the scientific committee of the NBB.
- When the Application is approved, the Researcher (‘Applicant’) is asked to provide the standard MTA to the person authorized to sign documents on behalf of the Legal Entity (e.g. board of directors), provided that no MTA has already been signed with that same Legal Entity. Formally the Legal Entity (called Recipient in the MTA) - not the individual researcher - is a party to the legal agreement. The responsibility to ensure compliance with the provisions of the MTA lies with the Legal Entity.
- When the Legal Entity has signed the MTA, details concerning Material Transfer (such as the amount of tissue provided, preservation mode and financial contribution) are specified in the Implementing Letter.
- All subsequent transfers to the Legal Entity only require signing of the Implementing Letter and optionally an MTS by the requesting scientist. Every Implementing Letter becomes an appendix to the existing MTA. Provisions of the framework MTA apply to every transfer within the same Legal Entity.
For more information on the material transfer procedure of the NBB, please read our brochure for tissue applicants.
Browse the e-NBB for samples Download the application form
HomeBrain tissueApplication-old
